483Feb 18, 2022
483 Document: Tissue Transplant Technology, Ltd. d/b/a Bone Bank Allografts
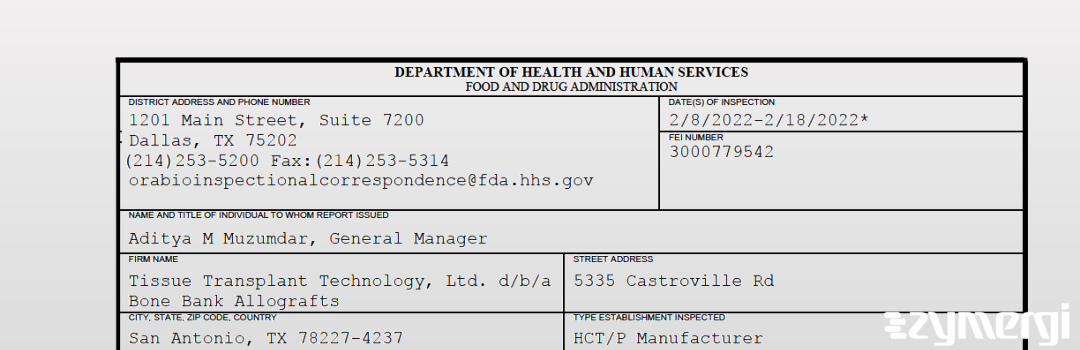
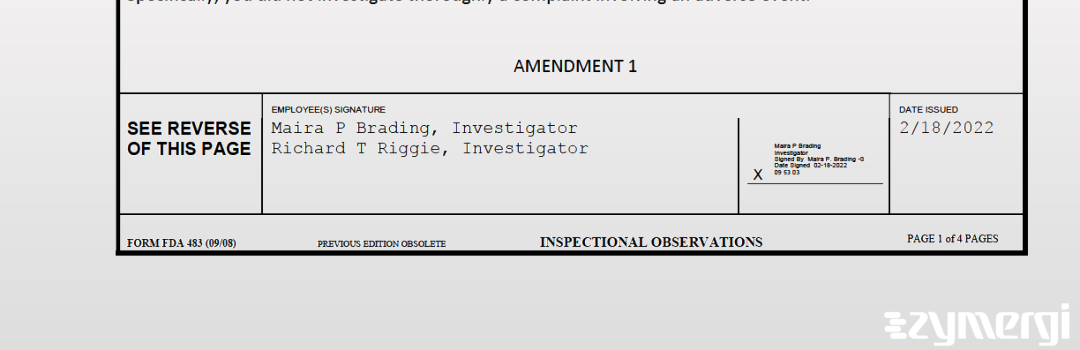
On Feb 18, 2022, the FDA inspected Tissue Transplant Technology, Ltd. d/b/a Bone Bank Allografts's San Antonio site. Explore the inspectional observations.


On Feb 18, 2022, the FDA inspected Tissue Transplant Technology, Ltd. d/b/a Bone Bank Allografts's San Antonio site. Explore the inspectional observations.