483Apr 14, 2023
483 Document: Par Sterile Products LLC
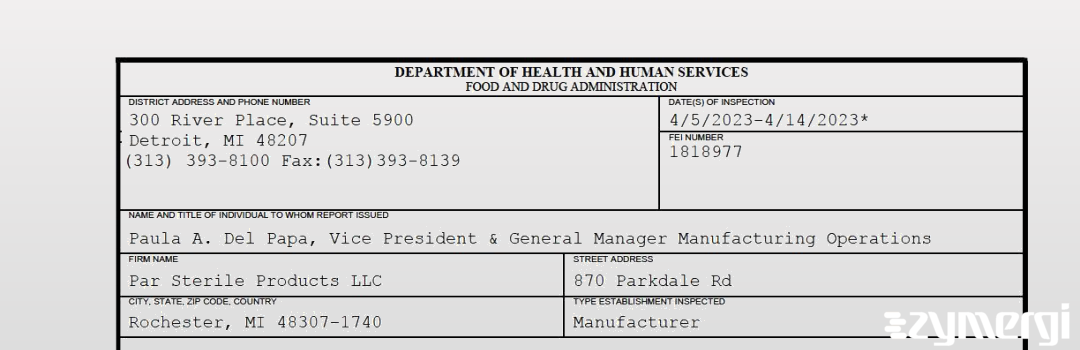
On Apr 14, 2023, the FDA inspected Par Sterile Products LLC's Rochester site. Explore the inspectional observations.


On Apr 14, 2023, the FDA inspected Par Sterile Products LLC's Rochester site. Explore the inspectional observations.