483Nov 17, 2022
483 Document: Hovione LLC
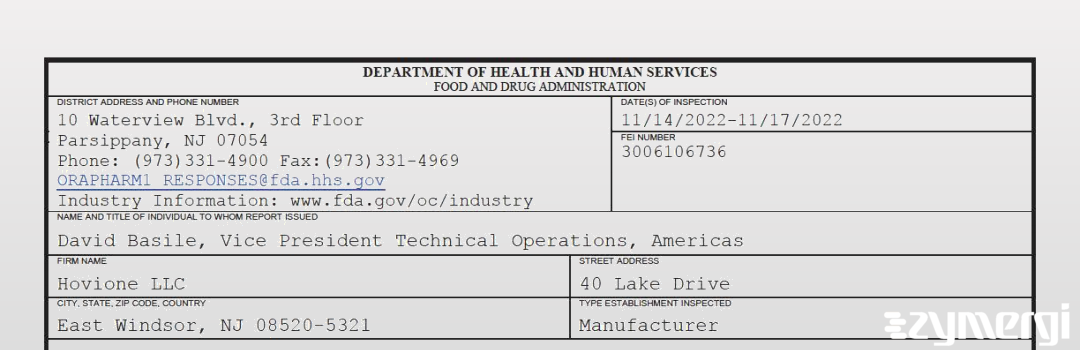

On Nov 17, 2022, the FDA inspected Hovione LLC's East Windsor site. Explore the inspectional observations.


On Nov 17, 2022, the FDA inspected Hovione LLC's East Windsor site. Explore the inspectional observations.