483Aug 10, 2023
483 Document: Cangene BioPharma, LLC
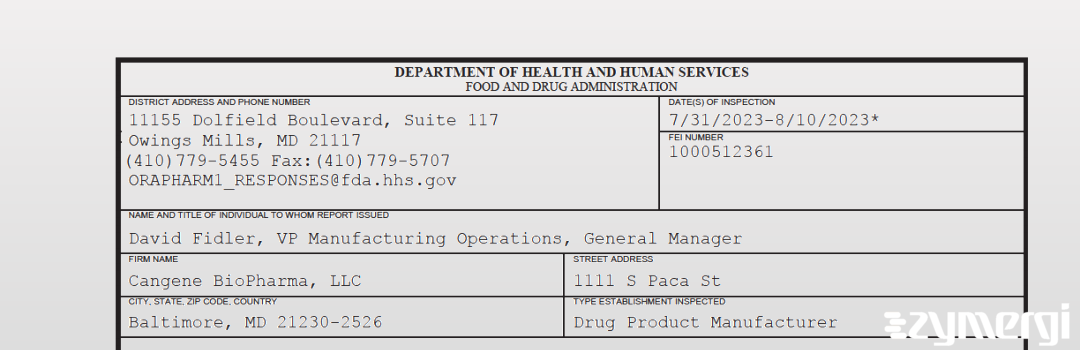
On Aug 10, 2023, the FDA inspected Cangene BioPharma, LLC's Baltimore site. Explore the inspectional observations.


On Aug 10, 2023, the FDA inspected Cangene BioPharma, LLC's Baltimore site. Explore the inspectional observations.